Comparative isotropy of the ¹³¹I-ChiTn radiolabeled monoclonal antibody in murine lung tumors treated with losartan
DOI:
https://doi.org/10.35954/SM2026.45.1.2.e301Keywords:
Antibodies, monoclonal, dosimetry, tumor microenvironment, radioimmunotherapy, ¹³¹I-ChiTnAbstract
Introduction: Radioimmunotherapy allows for the selective delivery of ionizing radiation to tumor cells using radiolabeled monoclonal antibodies. The anti-Tn chimeric antibody (ChiTn) radiolabeled with ¹³¹I represents a potential strategy for the targeted treatment of tumors that express this antigen.
Objective: To estimate the effect of pretreatment with losartan on the absorbed dose distribution of ¹³¹I-ChiTn in a murine model of Tn+ lung tumor.
Methodology: The absorbed dose was estimated by integrating time-activity curves derived from previously published biodistribution data and applying the MIRD formalism for internal dosimetry.
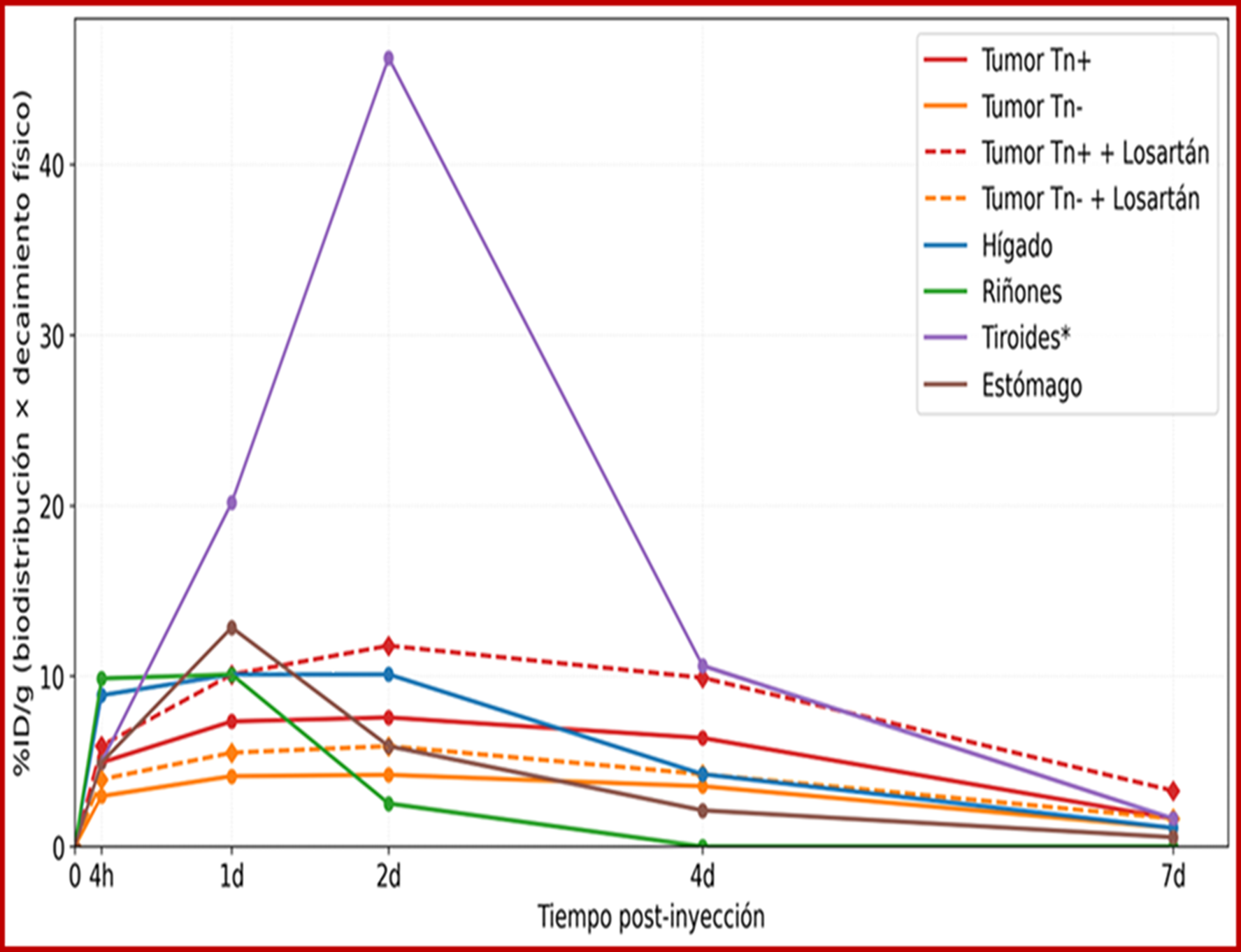
Results: Increased tumor uptake of the radiopharmaceutical in the presence of losartan, with an increase in the tumor absorbed dose from 1.5 to 1.8 mGy/MBq in Tn+ tumors, without significant increases in the dose to critical organs.
Discussion: These results suggest that pharmacological modulation of the tumor microenvironment using losartan could improve the therapeutic index of radioimmunotherapy with ¹³¹I-ChiTn and optimize the therapeutic efficacy of this radiolabeled antibody.
NOTE: This article has been approved by the Editorial Board.
Received for review: December 2025.
Accepted for publication: February 2026.
Publication date: March 2026.
Correspondence: Faculty of Sciences. Iguá 4225. Zip Code 11400. Tel.: (+598) 2525 8618. Montevideo, Uruguay.
Contact email: marcos.tassano@fcien.edu.uy
Downloads
Metrics
References
Parakh S, Lee ST, Gan HK, Scott AM. Radiolabeled antibodies for cancer imaging and therapy. Cancers (Basel). 2022 Mar 11; 14(6):1454. https://doi.org/10.3390/cancers14061454 PMID: 35326605; PMCID: PMC8946248. DOI: https://doi.org/10.3390/cancers14061454
Goldenberg DM. Targeted therapy of cancer with radiolabeled antibodies. J Nucl Med. 2002 May; 43(5):693-713. PMID: 11994535.
Jain RK. Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science. 2005 Jan 7; 307(5706):58-62 https://doi.org/10.1126/science.1104819 PMID: 15637262. DOI: https://doi.org/10.1126/science.1104819
Jain M, Venkatraman G, Batra SK. Optimization of radioimmunotherapy of solid tumors: biological impediments and their modulation. Clin Cancer Res. 2007 Mar 1; 13(5):1374-82. https://doi.org/10.1158/1078-0432.CCR-06-2436 Epub 2007 Feb 19. PMID: 17309914.
Chauhan VP, Martin J, Liu H, Lacorre D, Jain S, Kozin S, Stylianopoulos, T, Mousa A, Han X, Adstamongkonkul P, Popović Z, Huang P, Bawendi M, Boucher Y, Jain R. Angiotensin inhibition enhances drug delivery and potentiates chemotherapy by decompressing tumor blood vessels. Nat Commun. 2013; 4:2516. https://doi.org/10.1038/ncomms3516 DOI: https://doi.org/10.1038/ncomms3516
Munkley J. The role of sialyl-Tn in cancer. Int J Mol Sci. 2016 Feb 24; 17(3):275. https://doi.org/10.3390/ijms17030275 PMID: 26927062; PMCID: PMC4813139. DOI: https://doi.org/10.3390/ijms17030275
Ando H, Matsushita T, Wakitani M, Sato T, Kodama-Nishida S, Shibata K, Shitara K, Ohta S. Mouse-human chimeric anti-Tn IgG1 induced anti-tumor activity against Jurkat cells in vitro and in vivo. Biol Pharm Bull. 2008 Sep; 31(9):1739-44. https://doi.org/10.1248/bpb.31.1739 PMID: 18758069. DOI: https://doi.org/10.1248/bpb.31.1739
Tassano M, Camacho X, Freire T, Perroni C, da Costa V, Cabrera M, García MF, Fernandez M, Gambini JP, Cabral P, Osinaga E. Enhanced tumor targeting of radiolabeled mouse/human chimeric anti-Tn antibody in losartan-treated mice bearing Tn-expressing lung tumors. Cancer Biother Radiopharm. 2024 Jun; 39(5):337-348. https://doi.org/10.1089/cbr.2023.0138 Epub 2024 Jan 12. PMID: 38215243. DOI: https://doi.org/10.1089/cbr.2023.0138
Lundh C, Lindencrona U, Postgård P, Carlsson T, Nilsson M, Forssell-Aronsson E. Radiation-induced thyroid stunning: differential effects of 123I, 131I, 99mTc, and 211At on iodide transport and NIS mRNA expression in cultured thyroid cells. J Nucl Med. 2009; 50(7):1161–1167. https://doi.org/10.2967/jnumed.108.061150 DOI: https://doi.org/10.2967/jnumed.108.061150
Loevinger R, Budinger TF, Watson EE. MIRD Primer for absorbed dose calculations. New York, NY: Society of Nuclear Medicine, 1991.
Loke KS, Padhy AK, Ng DC, Goh AS, Divgi C. Dosimetric considerations in radioimmunotherapy and systemic radionuclide therapies: a review. World J Nucl Med. 2011 Jul; 10(2):122-38. https://doi.org/10.4103/1450-1147.89780 PMID: 22144871; PMCID: PMC3227338. DOI: https://doi.org/10.4103/1450-1147.89780
Vose JM, Wahl RL, Saleh M, Rohatiner AZ, Knox SJ, Radford JA, Zelenetz AD, Tidmarsh GF, Stagg RJ, Kaminski MS. Multicenter phase II study of iodine-131 tositumomab for chemotherapy-relapsed/refractory low-grade and transformed low-grade B-cell non-Hodgkin's lymphomas. J Clin Oncol. 2000 Mar; 18(6):1316-23. https://doi.org/10.1200/JCO.2000.18.6.1316 PMID: 10715303. DOI: https://doi.org/10.1200/JCO.2000.18.6.1316
Witzig TE. Yttrium-90-ibritumomab tiuxetan radioimmunotherapy: a new treatment approach for B-cell non-Hodgkin's lymphoma. Drugs of Today (Barcelona, Spain: 1998). 2004 Feb; 40(2):111-119. https://doi.org/10.1358/dot.2004.40.2.799423 PMID: 15045033. DOI: https://doi.org/10.1358/dot.2004.40.2.799423
Ray Banerjee S, Kumar V, Lisok A, Chen J, Minn I, Brummet M, Boinapally S, Cole M, Ngen E, Wharram B, Brayton C, Hobbs R, Pomper M. 177Lu-labeled low-molecular-weight agents for PSMA-targeted radiopharmaceutical therapy. Eur J Nucl Med Mol Imaging. 2019 November; 46(12): 2545–2557. https://doi.org/10.1007/s00259-019-04434-0 DOI: https://doi.org/10.1007/s00259-019-04434-0
Behr TM, Juweid ME, Sharkey RM, Dunn RM, Ying Z, Becker WS, Siegel JA, Goldenberg DM. Thyroid radiation doses during radioimmunotherapy of CEA-expressing tumours with 131I-labelled monoclonal antibodies. Nucl Med Commun. 1996 Sep; 17(9):767-80. https://doi.org/10.1097/00006231-199609000-00007 PMID: 8895904. 1996. DOI: https://doi.org/10.1097/00006231-199609000-00007
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Marcos Tassano, Pablo Cabral

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Until 2024 we use the Creative Commons Attribution/NonCommercial Attribution 4.0 International License https://creativecommons.org/licenses/by-nc/4.0/deed.es. Which states that: you are free to share, copy and redistribute the material in any medium or format, as well as to adapt, remix, transform and build upon the material. Under the following terms:
Attribution: you must give proper credit , provide a link to the license, and indicate if changes have been made . You may do so in any reasonable manner, but not in such a way as to suggest that you or your use is endorsed by the licensor.
NonCommercial: you may not use the material for commercial purposes.
As of 2025 authors retain their copyright and assign to the journal the right of first publication of their work, which shall simultaneously be subject to the license https://creativecommons.org/licenses/by-nc-sa/4.0/deed.es that permits sharing, copying and redistribution of the material in any medium or format provided that initial publication in this journal is indicated. Adapt, remix, transform and build upon the material. If you remix, transform, or build from the material, you must distribute your contribution under the same license as the original and may not make use of the material for commercial purposes.
Under the following terms:
1. Attribution: you must give proper credit, provide a link to the license, and indicate whether changes have been made. You may do so in any reasonable manner, but not in such a way as to suggest that you or your use is endorsed by the licensor.
2. NonCommercial: you may not use the material for commercial purposes.
3. ShareAlike: if you remix, transform or build upon the material, you must distribute your contribution under the same license as the original.
PlumX Metrics